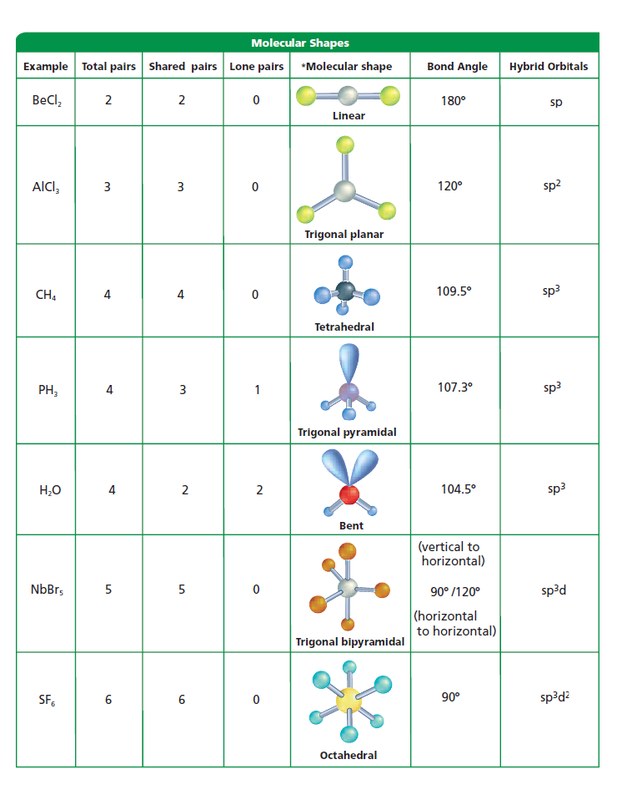
Thank you so much for asking a question, and i hope this was helpful. Why is PH3 trigonal pyramidal The PH3 molecule has one lone. That is our louis structure, our electron domain geometry and our molecular geometry. Looking at its Lewis structure we can state that molecular geometry of PH3 is trigonal pyramidal. Paramedele is a different shape, so you do typically need both of those words when describing this molecular geometry, all right. You do typically need both of these words for that, because trigonal can be used with plainer and that's a different shape and parameter could be used with square square. It'S a trigonal parameterall right, so that would be so boat. Rather, that is typically described using 2 words. This gives us an electron or molecular geometry. The 3 hydrogen is are going to be attached to an electron pair and then the last electron pair is just a lone pair. Well, the geometry of the molecule: it is going to have 3 atoms attached. We could take this step farther because it wants to know the molecular geometry. So the electron domain geometry is a tetrahedral, because we have 4 electron domains attached to that central atom and according to vesper theory, they will arrange themselves as far away from each other as possible. The correct Lewis structure for BF3 would. 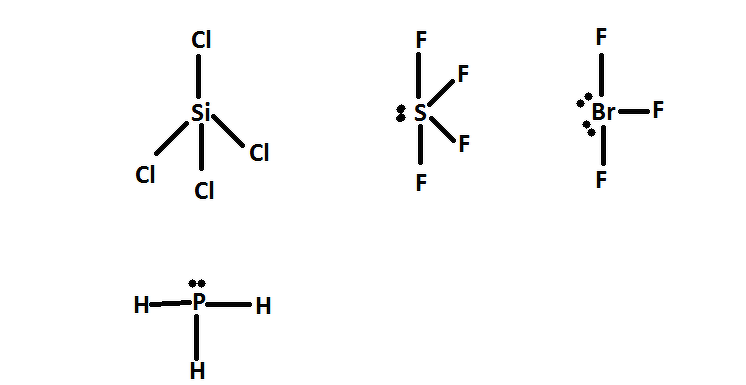
If we have our central atom here- and it has 4 electron domains on it, that is going to give us an electron domain geometry that is a tetrahedral. What is the molecular geometry of PH3 (A) Trigonal planar (B) T-shaped (C) Trigonal pyramidal (D) Tetrahedral 17. There are 3 atoms bonded to it and it has 1 lone pair, adding together the number of bonded atoms and the number of lone pairs gives us a total of 4, and that means 4 electron domains. Journal of the Chemical Society, Faraday Transactions 2: Molecular and Chemical Physics. 
Well, let's look at our central atom or central atoms phosphorus. The geometries of PH, PF and their ground ionic states have been. Our next question, then, is the electron domain geometry. 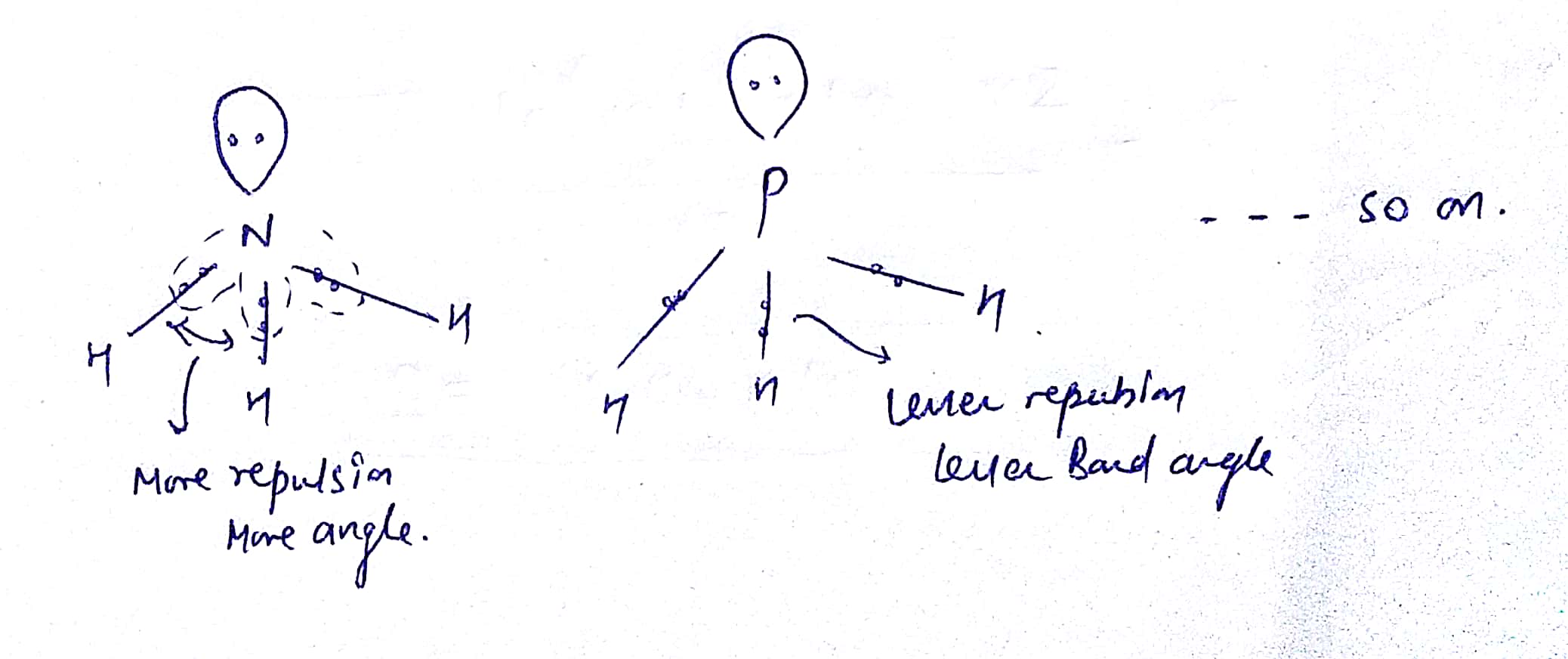
We'Ve used exactly 8 dots and dictet rule has been satisfied. Hydrogen has a full first energy level with just 2 electrons phosphorus, however, needs 2 more to satisfy the octet rule, so we're going to put a lone pair of electrons on the phosphorus, and this is our completed. Each hydrogen already has its necessary electrons. Each hydrogen has to be bonded to the phosphorus that is going to require a pair of electrons for each of those, and that uses up 6 of our electrons. Let'S go ahead and draw our skeleton structure for the skeleton structure we put the single atom in the center and we're going to surround it by the 3 hydrogen. Draw the Lewis structure of PH3 molecule and state its electron domain geometry. Therefore, that means we need 8 dots all right. Valence electrons are represented as dots. the reactivity of a molecule and how it might interact with other molecules. What is pH3's molecular shape The question makes no sense, but p orbitals are two lobes in each of the 3 dimensions, one lobe being geometrically positive, one negative. Due to greater lone pair-bond pair repulsion than bond pair-bond pair repulsion, the tetrahedral angle. Adding these together gives us 8 valence electrons in a lewis structure. We draw Lewis Structures to predict: -the shape of a molecule. But PH3 has three bond pairs and one lone pair around P. Because there are four electrons regions, geometry is tetrahedral and shape is trigonal pyramidal. So, hybridization of phosphorus atom is sp 3. Therefore, there are total of four electrons regions. There are 3 of them and each hydrogen is in group 1 of the periodic table, meaning they each have 1 balance electron. Lewis structure of Phosphine molecule contains three sigma bonds and one lone pair around phosphorus atom. Therefore it has 5 valence electrons hydrogen. So phosphorus is in group 5 of the periodic table. We need to know the total number of valence electrons that we are working with. The result can be described as a distorted tetrahedron around boron.Hi there in this question we have the molecule ph 3, and the first thing we want to do is to draw its lewis structure to draw louis structure. $\ce$ banana bonds of the 2-electron-3-centre type.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
